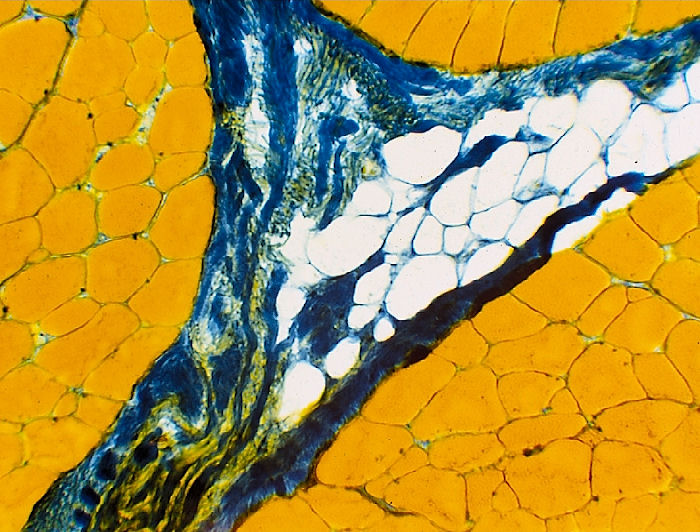

Aniline Blue - Orange G Stain
This stain provides a
good contrast between muscle fibers (orange), connective tissue (blue), and
fat cells (white).

1. Cut
tissue sample on a microtome 10 to 12 µm thick and place onto microscope
slides (microscope slides should be pre-rinsed/cleaned in an ethanol
solution).
Leave slides at room temperature for at least 5 min.
2. Fix sections using Müller´s reagent for 1 min.
3. Rinse for 1 to 2 min in d2H2O.
4. Stain sections with Orange G for 3 min.
5. Rinse for 2 min in dH2O (twice).
6. Stain sections with Aniline Blue for 1 to 3 seconds.
7. Rinse (with gentle shaking) immediately for 2 to 5 minutes in dH2O
(twice) (until no blue color is seen in the water)
8. Wash with:
70% Ethanol for 5 to 10 sec
96% Ethanol for 5 to 10 sec
absolute Ethanol for 1 to 2 min
9. Fix with Xylol for between 5 to 60 min (dispose of in proper receptacle).
10. Mount a cover slip using a non-aqueous cover slip medium (i.e.
Histofluid).
Solutions and Reagents:
Müller´s reagent (Fresh Daily)
Potassium dichromate (K2Cr2O7)
2.5 g
Sodium
sulfate (Na2SO4-10H2O) 1.0
g
dH2O
100 mL
Phenol
pinch (tip of a scupula, see notes)
Orange G solution (Store at room temperature, good for over 1 year)
Orange G
3.0 g
Acetic Acid, conc.
5.0
mL
dH2O
100 mL
Bring the solution to a boil, cool to room temperature, and filter.
Aniline Blue
0.5 g
Acetic Acid, conc.
5.0 mL
dH2O
100 mL
Bring the solution to a boil, cool to room temperature, and filter.
Notes:
- The
phenol added to the aniline blue/orange G stain is used as a preservative to
stop bacterial growth. The recipe is over 100 years old and was used when
water sources were not as clean as they are today. Imagine the end of a pen
and use an amount of phenol equivalent to the tip of the pen. Its basically
just a few grains.